These Genetically Engineered Brain Cells Devour Toxic Alzheimer’s Plaques
Scientists Transform Brain Cells into Amyloid-Gobbling Machines in Breakthrough Alzheimer’s Research
Alzheimer’s disease and cancer share a frustrating similarity: both have defied decades of scientific effort to find reliable treatments. While cancer research has seen revolutionary advances through CAR T cell therapy, Alzheimer’s research has remained frustratingly stagnant—until now.
A groundbreaking study from Washington University School of Medicine in St. Louis has adapted CAR T technology to create something entirely new: CAR-A, or chimeric antigen receptor astrocytes. This innovative approach transforms the brain’s support cells into specialized amyloid-clearing machines, potentially offering a lasting solution to the devastating disease.
The Alzheimer’s Puzzle: Why Treatment Has Been So Difficult
For over a century, Alzheimer’s has baffled scientists. The disease gradually destroys memory, decision-making, and movement by causing neurons to wither. At the center of this destruction are amyloid proteins, which form toxic clumps in the brain.
The relationship between amyloid and Alzheimer’s remains hotly debated. Some researchers argue these protein clumps are the primary cause, while others believe they’re merely a symptom of deeper problems. What we do know is that in Alzheimer’s patients, amyloid proteins clump together in ways that overwhelm the brain’s natural cleanup systems.
Current treatments focus on clearing these amyloid plaques using antibody drugs. However, these medications require regular administration and carry significant risks, including brain bleeds and stroke-like symptoms. Many patients opt out of treatment due to these dangers, leaving millions without effective options.
Borrowing from Cancer’s Success Story
The inspiration for this new approach comes from one of medicine’s most remarkable success stories: CAR T cell therapy for blood cancers. This treatment works by genetically engineering a patient’s T cells to recognize and destroy cancer cells. The engineered cells produce special proteins called chimeric antigen receptors (CARs) that can lock onto specific targets.
The Washington University team wondered: could this same principle work against amyloid proteins in the brain?
The Birth of CAR-A: Engineering Astrocytes to Fight Alzheimer’s
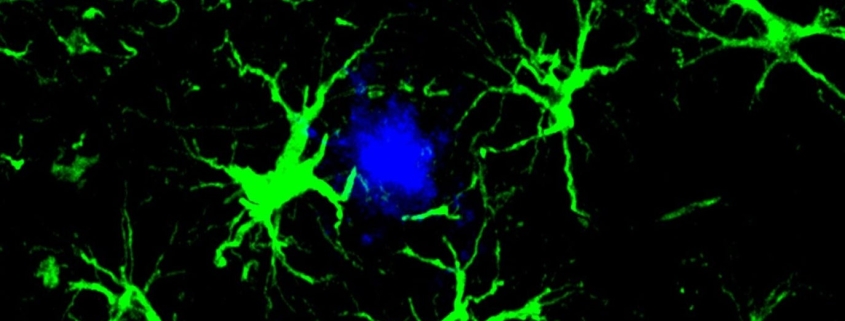
The researchers turned to astrocytes, the brain’s most abundant cell type. These star-shaped support cells help repair brain injuries, provide nutrients to neurons, and play crucial roles in memory and cognition. While astrocytes can naturally clear some proteins, they’re not particularly efficient at removing amyloid.
To boost their amyloid-clearing abilities, the team tested multiple CAR designs. They packaged the genetic instructions for the most promising designs into harmless viruses and injected them into mice modeling Alzheimer’s disease.
Once inside the brain, these viruses transformed naturally occurring astrocytes into CAR-A cells—astrocytes equipped with the ability to seek out and destroy amyloid proteins.
Results That Could Change Everything
The results were remarkable. In young mice (roughly equivalent to human teenagers), a single injection prevented amyloid clump formation for at least three months. In older mice already suffering from amyloid buildup, the treatment cut amyloid levels by approximately 50%.
But the benefits extended beyond just clearing amyloid. The CAR-A cells also triggered the brain’s immune cells to more actively devour amyloid plaques, swept away malfunctioning immune cells, and reduced overall brain inflammation.
This multi-pronged approach could create a less toxic environment in the Alzheimer’s brain compared to current treatments that focus solely on amyloid clearance.
The Blood-Brain Barrier Advantage
One of the biggest challenges in treating brain diseases is the blood-brain barrier—a protective shield that prevents many drugs from reaching their targets. Anti-amyloid antibodies in current Alzheimer’s drugs struggle to penetrate this barrier effectively.
CAR-A cells, however, are produced directly inside the brain, potentially offering better access to their targets with minimal damage to blood vessels. This could significantly reduce the risk of deadly side effects that plague current treatments.
Early Intervention Shows Most Promise
As with antibody treatments, the CAR-A approach proved most effective when administered early in the disease process. This aligns with growing evidence that Alzheimer’s treatments work best before extensive damage occurs.
Study author David Holtzman noted the key advantage: “Where it differs, and where it could make a difference in clinical care, is in the single injection that successfully reduced the amount of harmful brain proteins in mice.”
The Same Old Problem Persists
Despite the impressive reduction in amyloid levels, the treatment ran into a familiar obstacle: it didn’t significantly improve memory or mood in the mice. This mirrors results from anti-amyloid drug trials and highlights a fundamental question in Alzheimer’s research.
Why doesn’t clearing amyloid lead to better symptoms? Several theories exist. The CAR-A cells might be too aggressive, accidentally damaging neurons and their connections. More fundamentally, amyloid might not be the primary driver of symptoms after all.
Beyond Amyloid: The Tau Problem
As Alzheimer’s progresses, another protein called tau begins to aggregate in neurons. Higher tau levels correlate strongly with mental decline, suggesting that targeting amyloid alone might be insufficient.
Early treatments targeting tau have universally failed in clinical trials, but the CAR-A platform could be redesigned to go after both amyloid and tau simultaneously. This combination approach might finally break through the treatment barrier that has frustrated researchers for decades.
Future Applications Beyond Alzheimer’s
The potential applications extend far beyond Alzheimer’s disease. The same strategy could potentially target other neurodegenerative disorders or even brain cancers. The research team is already working on fine-tuning their designs to better detect various targets.
As CAR technologies mature and our ability to selectively neutralize toxic proteins improves, these approaches hold substantial promise for Alzheimer’s disease and other neurodegenerative disorders, according to researchers Jake Boles and David Gate from Northwestern University.
Looking Forward
While this research represents a significant breakthrough, much work remains before CAR-A therapy could become available to patients. The technology must be refined, safety must be established in larger animal models, and eventually, human trials would be necessary.
However, this innovative approach—borrowing successful cancer technology and adapting it for brain diseases—offers new hope in the long battle against Alzheimer’s. By transforming the brain’s own cells into disease-fighting machines, scientists may have found a way to work with the body rather than against it.
The journey to effective Alzheimer’s treatment has been long and frustrating, but this research suggests that the answer might come from unexpected places—in this case, from the same technology that revolutionized blood cancer treatment.
Tags: Alzheimer’s breakthrough, CAR-A therapy, brain cell engineering, amyloid clearance, Washington University research, neurodegenerative disease treatment, CAR T technology, astrocytes, dementia research, medical innovation
Viral phrases: “brain cells transformed into amyloid-gobbling machines,” “revolutionary approach to Alzheimer’s treatment,” “CAR-A cells cut amyloid by 50%,” “single injection prevents plaque formation,” “borrowing cancer technology for brain disease,” “the blood-brain barrier breakthrough,” “engineering the brain’s support cells,” “multi-pronged attack on Alzheimer’s,” “targeting both amyloid and tau,” “working with the body’s own cells”
,


Leave a Reply
Want to join the discussion?Feel free to contribute!