Scientists Uncover the Hidden Mechanism Behind FDA-Approved Alzheimer’s Drug
Scientists Uncover the Hidden Mechanism Behind FDA-Approved Alzheimer’s Drug
In a groundbreaking discovery that could reshape the future of Alzheimer’s treatment, researchers have finally unlocked the secret behind how lecanemab, the FDA-approved monoclonal antibody therapy marketed as Leqembi, actually works to clear toxic amyloid plaques from the brain. This revelation, published by scientists from VIB and KU Leuven, represents a significant leap forward in our understanding of Alzheimer’s disease and offers new hope for millions of patients worldwide.
Alzheimer’s disease, a progressive neurodegenerative disorder, has long been characterized by the accumulation of amyloid-beta plaques and tau tangles in the brain. These toxic protein aggregates disrupt neural communication and lead to the gradual deterioration of cognitive function. For decades, researchers have been searching for effective treatments that could halt or reverse this devastating process.
Enter lecanemab, a humanized monoclonal antibody that targets amyloid-beta. Since its FDA approval in 2023, the drug has shown promise in reducing amyloid plaque burden and slowing cognitive decline in clinical trials. However, the exact mechanism by which lecanemab exerts its therapeutic effect remained a mystery – until now.
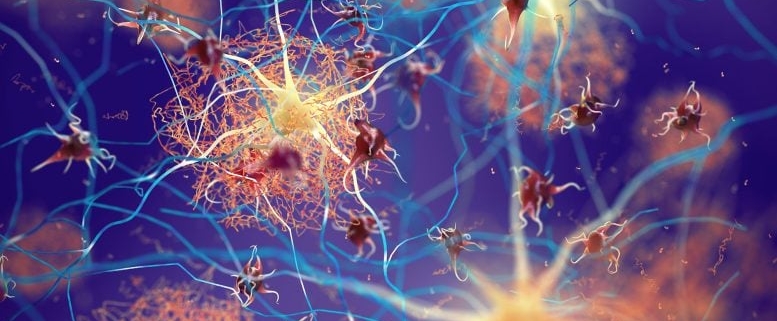
The research team, led by Dr. Bart De Strooper and Dr. Mark Fiers, has discovered that lecanemab’s effectiveness hinges on its ability to engage microglia, the brain’s resident immune cells, through its Fc fragment. This finding is crucial because it not only explains how the drug works but also opens up new avenues for developing more targeted and effective Alzheimer’s treatments.
Microglia play a vital role in maintaining brain health by clearing debris, fighting infections, and regulating inflammation. In Alzheimer’s disease, these cells often become dysfunctional, contributing to the progression of the disease. The researchers found that when lecanemab binds to amyloid-beta plaques, its Fc fragment interacts with Fc receptors on microglia, triggering a cascade of cellular events that lead to the clearance of the plaques.
This discovery is particularly significant because it defines the cellular program behind lecanemab’s therapeutic effect. By understanding this mechanism, scientists can now explore ways to enhance or modify the drug’s action, potentially leading to more effective treatments with fewer side effects.
The implications of this research extend far beyond lecanemab itself. It provides a framework for understanding how other antibody-based therapies might work in the brain and could guide the development of next-generation treatments for Alzheimer’s and other neurodegenerative diseases.
Moreover, this finding underscores the importance of considering the role of the immune system in neurodegenerative disorders. It suggests that modulating immune responses in the brain could be a key strategy in treating these conditions, opening up new research directions and potential therapeutic approaches.
The study, published in the prestigious journal Nature Neuroscience, is the result of years of collaborative effort involving advanced imaging techniques, genetic analysis, and in-depth cellular studies. The researchers used state-of-the-art methods to track the movement and activity of lecanemab in the brain, as well as to analyze the response of microglia at a molecular level.
One of the most exciting aspects of this discovery is its potential to inform personalized medicine approaches to Alzheimer’s treatment. By understanding the specific cellular mechanisms involved, doctors may be able to better predict which patients are most likely to respond to lecanemab or similar therapies, allowing for more tailored treatment plans.
However, the researchers caution that while this discovery is a significant step forward, there is still much work to be done. Alzheimer’s disease is complex and multifaceted, and amyloid plaques are just one piece of the puzzle. Future research will need to explore how lecanemab interacts with other aspects of the disease, such as tau tangles and neuroinflammation, to provide a more comprehensive understanding of its therapeutic potential.
The team is already planning follow-up studies to investigate how the drug’s mechanism of action might vary in different stages of Alzheimer’s disease and in patients with varying genetic backgrounds. They are also exploring whether similar mechanisms could be harnessed to develop treatments for other neurodegenerative disorders.
As the global population ages, the need for effective Alzheimer’s treatments becomes increasingly urgent. With over 6 million Americans currently living with Alzheimer’s and that number expected to double by 2050, breakthroughs like this offer a glimmer of hope in the fight against this devastating disease.
The discovery of lecanemab’s mechanism of action represents a significant milestone in Alzheimer’s research. It not only provides a deeper understanding of how this promising therapy works but also opens up new avenues for developing more effective treatments. As research in this field continues to advance, we move closer to a future where Alzheimer’s disease may no longer be an inevitable consequence of aging but a manageable condition with effective treatments available.
#TagsAndViralPhrases
#AlzheimersBreakthrough #LecanemabMechanism #BrainHealthRevolution #NeurodegenerationSolved #MicrogliaMagic #AmyloidClearance #CognitiveDeclineStopped #LeqembiWorks #AlzheimersResearch #BrainImmunity #NeuroScienceNews #DementiaTreatment #MonoclonalAntibodySuccess #VIBKULeuvenDiscovery #NatureNeuroscience #AlzheimersHope #BrainPlaqueRemoval #ImmunotherapyForAlzheimers #NeurodegenerativeDiseaseResearch #AlzheimersTreatmentFuture #BrainHealthInnovation #CognitivePreservation #NeuroScienceAdvancement #AlzheimersTherapyMechanism #BrainImmuneSystem #NeuroPlaqueClearing #AlzheimersDrugMechanism #BrainHealthBreakthrough #NeurodegenerationResearch #AlzheimersHopeForMillions #BrainHealthScience #NeuroScienceDiscovery #AlzheimersTreatmentAdvancement #BrainImmunityResearch #NeuroPlaqueRemoval #AlzheimersResearchBreakthrough #BrainHealthTreatment #NeurodegenerativeDiseaseHope #AlzheimersTherapySuccess #BrainImmuneResponse #NeuroScienceInnovation #AlzheimersDiseaseResearch #BrainHealthAdvancement #NeurodegenerationTreatment #AlzheimersHopeResearch #BrainImmunityDiscovery #NeuroScienceProgress #AlzheimersTreatmentHope #BrainHealthScienceNews #NeurodegenerativeDiseaseBreakthrough,



Leave a Reply
Want to join the discussion?Feel free to contribute!